 Bohr used his model to explain the spectral lines of hydrogen. 
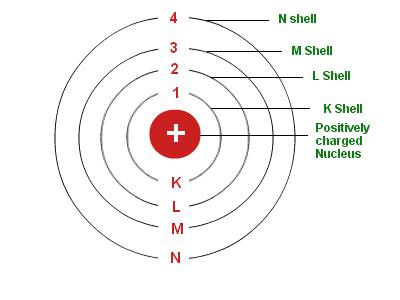
This atomic model was the first to use quantum theory, in that the electrons were limited to specific orbits around the nucleus. The development of quantum mechanics in the 1920s revolutionized our understanding. Niels Bohr developed the planetary model of the atom in 1913. Ernest Rutherfords gold foil experiment in 1911 led to the discovery of the atomic nucleus. This means that electrons’ energy levels are discrete and that the electrons revolve in stable orbits around the nucleus, but can jump from one energy level to another. Niels Bohr - Atomic Theory, Quantum Mechanics, Nobel Prize: After the discovery of fission, Bohr was acutely aware of the theoretical possibility of making an atomic bomb. John Dalton proposed the first modern atomic theory in 1803. As part of his contributions, Bohr developed the ‘Bohr Model’ of the atom, in which he proposed that the electron was able to occupy only certain orbits around the atomic nucleus. An important contribution to the problem of a consistent application of these. Bohr was able to discover the mechanism of internal functioning. It was the first to exhibit the atom as a positively charged nucleus and surrounded by orbiting electrons. At the university, he came under the guidance of Prof C Christiansen, and earned his Master’s degree in Physics in 1909 and Doctorate in 1911. The atomic model discovered by Niels Bohr is considered one of his greatest contributions to the world of physics and the sciences in general. In 1903, Bohr enrolled as an undergraduate at the University of Copenhagen, majoring in physics, while also studying astronomy and mathematics. One of his best-known students was Ernest Rutherford, who succeeded Thomson as Cavendish Professor of Physics.Bohr was born in Copenhagen on October 7, 1885, the second of three children of Christian Bohr, a professor of physiology at the University of Copenhagen, and his wife Ellen. Seven of his research assistants, as well as his own son, went on to win the Nobel Prize in Physics. Thomson's greatest contribution to science to be his role as a teacher. Thomson published an important monograph in 1913 urging the use of the mass spectrograph in chemical analysis. His atomic theory helped explain atomic bonding and the structure of molecules. Thomson was closely aligned with chemists of the time. Please be aware that some material may contain words, descriptions or illustrations which will not reflect current scientific understanding and may be.These experiments led to the development of the mass spectrograph. He worked on the Manhattan Project. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
